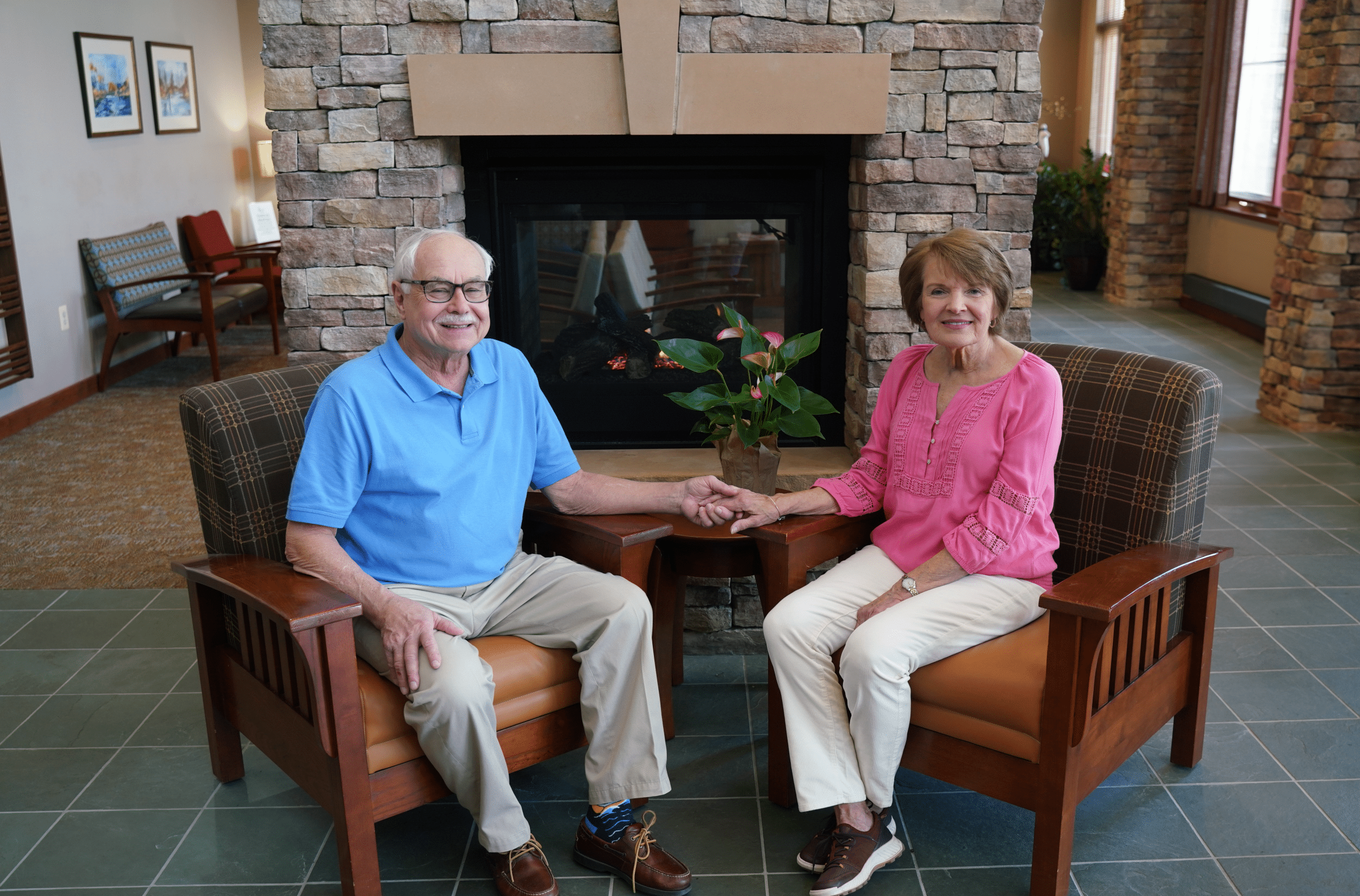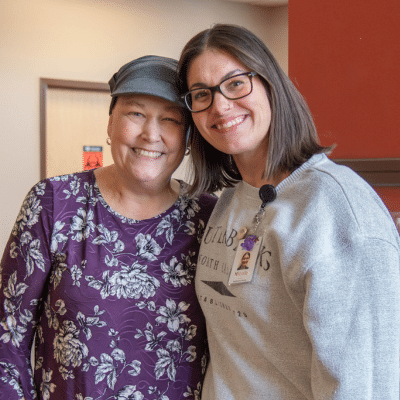Where Healing Begins, Strength Prevails
The HOAF Difference
At HOAF, our team is committed to delivering exceptional cancer and blood disorder care for the Fredericksburg, Stafford and surrounding communities. Our practice combines leading-edge treatments with a personalized touch to provide top-tier services. As an independently-owned practice, we prioritize personalized care, ensuring that each patient receives comprehensive and compassionate care tailored to their specific needs. Our state-of-the-art facility with an on-site pharmacy and lab ensures convenience and streamlines your treatment experience. When you choose HOAF for your partner in health, you are receiving holistic care that is designed with your comfort and healing in mind.
our mission
We believe that clinical and warm are not mutually exclusive. We believe that cancer is personal—for our patients and ourselves. We believe that a life spent caring for others at their most vulnerable point is the only life worth living. We believe that love and leading-edge treatment change and save lives. We believe that without passion, life is nothing.
Our passion is our patients.
Supporting Every Step In Your Fight
meet our compassionate team
your home for healing at our Fredericksburg and Stafford locations
our services
We are committed to delivering exceptional care for blood disorders and cancer. Our team of expert medical oncologists, skilled providers, and compassionate registered nurses is dedicated to making your experience comfortable and positive. With three convenient locations, our state-of-the-art facilities are designed with patient comfort in mind and feature the latest technologies for an exceptional treatment experience. We offer a wide range of services tailored to meet all your healthcare needs, including hematology, medical oncology, infusions, side effect management, a surveillance clinic, and on-site prescription services.
symptom relief in Fredericksburg & Stafford
side effect management
We are dedicated to helping our patients feel their best throughout their treatment journey. While treatments such as chemotherapy may bring unwanted side effects, our team is here to help you manage them. From the comfort and safety of our offices in Fredericksburg and Stafford, we address these symptoms to ensure our patients can navigate them with confidence and find relief.
acute care when you need it
surveillance clinic in fredericksburg
Our goal is to help patients avoid hospitalization whenever possible. We manage all aspects of care during treatment, addressing everything from the flu to mental health support through our Supportive Care Clinic at The Pines in Fredericksburg.
A Hand To Hold And To Heal
patient stories
We believe in the power of patient stories and the experiences shared by those who have navigated their cancer journey with us. Here, you can explore personal testimonials that reflect the compassionate, individualized care at the heart of our practice. These reviews offer insight into the support, expertise, and commitment Hematology Oncology Associates of Fredericksburg brings to every step of your cancer care.
Leading With Compassion, Treating With Expertise In Fredericksburg and Stafford, VA
Request an appointment
"*" indicates required fields